Dendritic cells (DCs), as professional antigen-presenting cells, play an indispensable role in initiating anti-tumor immune responses. Among the various DC subsets, type 1 conventional dendritic cells (cDC1s), with their exceptional ability to cross-present tumor antigens and activate CD8⁺ T cells, serve as a central hub in anti-tumor immunity. However, within the tumor microenvironment, tumor-derived metabolites and microenvironmental signals often inhibit cDC1 function, leading to impaired antigen presentation and the induction of immune tolerance. Therefore, in-depth elucidation of the molecular network regulating cDC1 function and its interaction with the tumor microenvironment is of great significance for optimizing cancer immunotherapeutic strategies.
Recently, the groups of Zuliang Jie from Xiamen University, Xun Li from the First Affiliated Hospital of Xiamen University, and Jian-Hong Shi from the Affiliated Hospital of Hebei University published a research paper titled "DYRK1A enhances antitumor immunity in type 1 conventional dendritic cells via mTORC1 activation" in The Journal of Clinical Investigation. This study identified the critical role of the kinase DYRK1A in regulating cDC1 function and systematically elucidated its molecular mechanism and potential clinical therapeutic implications.

The study found that DYRK1A is highly expressed in tumor-infiltrating DCs within the tumor microenvironment, and its expression levels can be significantly induced by TLR ligands, nutrients, and T cell-derived signals (such as IFN-γ). Using a cDC1-specific Dyrk1a knockout mouse model, the authors demonstrated that DYRK1A deficiency significantly accelerates tumor growth, accompanied by impaired proliferation of tumor-infiltrating CD8⁺ T cells, upregulation of exhaustion markers (PD-1, TIM-3), and decreased secretion of effector cytokines. Transcriptomic and flow cytometric analyses revealed that DYRK1A deficiency specifically downregulates the expression of MHC class I molecules and co-stimulatory molecules on the surface of cDC1s, with no significant effect on the cDC2 subset. Further analysis of antigen processing revealed that DYRK1A deficiency promotes lysosomal biogenesis and phagosome acidification in cDC1s, leading to excessive degradation of exogenous antigens within lysosomes and thereby severely impairing proteasome-dependent antigen cross-presentation.
Mechanistic studies indicated that DYRK1A promotes cDC1 maturation and activation by regulating the TSC2-mTORC1 signaling axis. DYRK1A directly binds to TSC2 and specifically phosphorylates Ser540. This phosphorylation enhances TSC2 binding to the E3 ubiquitin ligase HUWE1, thereby inducing ubiquitination and proteasomal degradation of TSC2 and relieving its inhibitory effect on mTORC1. Genetic experiments showed that concurrent Tsc2 knockout in Dyrk1a-deficient cDC1s completely restores mTORC1 activation and the antigen-presenting function of cDC1s. Furthermore, introducing a phosphomimetic TSC2 mutant (S540D) was sufficient to reshape the potent activation state of cDC1s. These results confirm that TSC2 is a central mediator in this regulatory axis and that phosphorylation at Ser540 is critical for maintaining mTORC1 activity and cDC1 function.
At the functional level, maintaining the activity of the DYRK1A-TSC2 axis significantly enhances the anti-tumor immune response of cDC1s. In a cDC1-based adoptive cell therapy model, Tsc2 knockout successfully rescued the immunogenic defects of DYRK1A-deficient cDC1s, restoring intratumoral effector T cell infiltration and tumor control ability. In TCGA and single-cell sequencing data from multiple human cancer types (e.g., melanoma, bladder cancer, breast cancer, and lung cancer), high DYRK1A expression in cDC1s was positively correlated with activation of mTORC signaling, enrichment of effector T cell signatures, and improved overall patient survival. These findings suggest that intervening in the DYRK1A-TSC2 signaling axis holds potential for clinical translation.
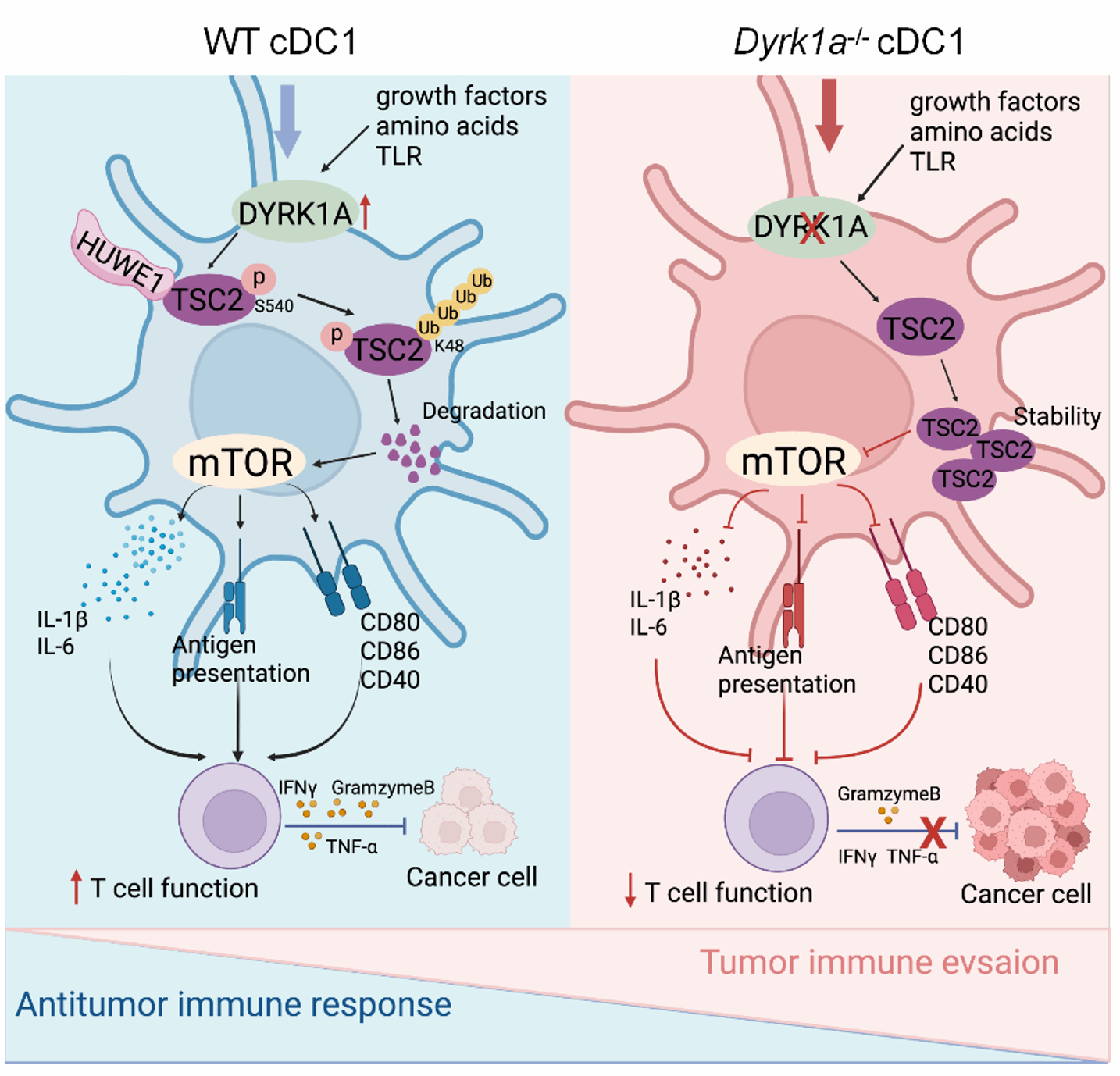
In summary, this study identifies a new "switch" for cDC1 anti-tumor immunity—DYRK1A. As a key microenvironmental signal-sensing kinase, DYRK1A promotes TSC2 degradation via phosphorylation, thereby activating the mTORC1 signaling pathway, optimizing antigen processing and cross-presentation by cDC1s, and playing a core regulatory role in anti-tumor immunity. Targeting DYRK1A activation or inhibiting TSC2 function may offer new strategies to enhance the efficacy of DC vaccines and cDC1-related cancer immunotherapies.
Professor Jie Zuliang from the School of Life Sciences, Xiamen University, Associate Professor Li Xun from the First Affiliated Hospital of Xiamen University, and Professor Shi Jianhong from the Affiliated Hospital of Hebei University are the co-corresponding authors of this study. Postdoctoral fellow Wang Hongjiao, master's students Jiang He and He Songlin, and postdoctoral fellow Ren Songwen from the School of Life Sciences, Xiamen University, are the co-first authors. Master's student Zhou Chunyun from the School of Public Health, Xiamen University, master's student Zhu Pan, and undergraduate student Chen Keren from the School of Life Sciences made significant contributions to this research. This study was supported by the National Key Research and Development Program of China, the National Natural Science Foundation of China, the Key Healthcare Projects of Xiamen City, and the scientific research foundation of state key laboratory of vaccines for infectious diseases, Xiang’an biomedicine laboratory.
Link to the original article: https://www.jci.org/articles/view/199108