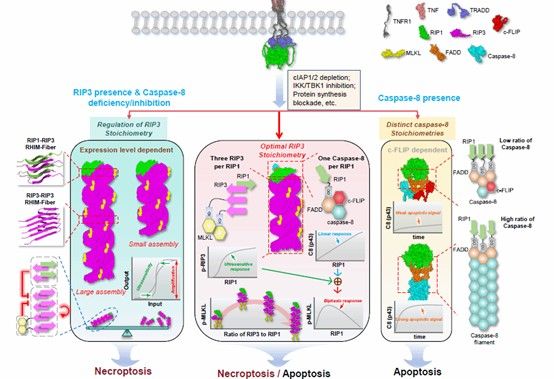
Abstract: Necrosome assembly is essential for necroptosis, a process implicated in neurodegeneration, ischemic injury, and inflammatory diseases. Yet the spatiotemporal rules governing this assembly remain elusive. Leveraging quantitative STORM and mathematical modeling, we define an approximately 3:1 ratio of RIP3 to RIP1 in necrosomes as the optimal stoichiometry for necroptosis, enabling signal amplification and a threshold response. Surprisingly, excessive RIP3 oligomerization attenuates signaling, acting as an intrinsic size control mechanism. RIP3 assembly is dynamically regulated: it is constrained by stimulation and RIP1, promoted by RIP3 itself, and unexpectedly limited by downstream MLKL. A complementary balance between necrosome quantity and RIP3 assembly degree ensures efficient MLKL phosphorylation. In contrast, Caspase-8 assembly is limited by c-FLIP and recruited linearly by RIP1, while its distinct behavior from RIP3 underlies the biphasic necroptotic response to RIP1. These findings uncover the flexible, multi-strategic nature of signalosomes and offer valuable insights for therapeutic and synthetic biology.
Link: https://www.nature.com/articles/s41467-025-67098-5