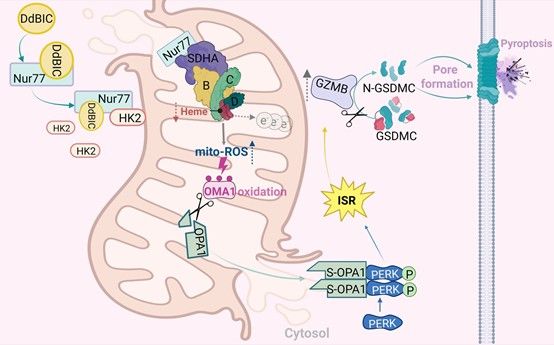
Abstract: Pyroptosis plays a crucial role in physiological and pathological processes. As melanoma cells are resistant to apoptosis but express gasdermin proteins, it is appealing to counter melanoma with the induction of gasdermin-executed pyroptosis. GSDMC, initially cloned from metastatic melanoma cells, has been demonstrated as a potential executioner of pyroptosis. However, no lead compounds that trigger GSDMC-mediated pyroptosis have been reported, which limits the in-depth investigation of GSDMC functions. Here, we discovered a chemical compound, dodecyl 1H-benzo[d]imidazole-5-carboxylate (DdBIC), that targeted the nuclear receptor Nur77 to induce pyroptosis through cleaving GSDMC by granzyme B in melanoma cells. Upon DdBIC binding, Nur77 was translocated to the mitochondria to activate the hemoprotein SDHA to overconsume succinyl-CoA, subsequently disrupting the homeostasis of heme in the SDH complex and resulting in electron leakage to induce mito-ROS production. This mito-ROS signal was sensed by the mitochondrial protease OMA1 via oxidation, which led to downstream OPA1 cleavage and subsequent released into the cytoplasm. Cytosolic OPA1 activated PERK to induce the integrated stress response (ISR), which further activated granzyme B to cleave GSDMC, culminating in the induction of pyroptosis. Together, this study elucidates a signal cascade from Nur77-impaired homeostasis of heme metabolism to PERK-mediated ISR activation, and reveals a novel paradigm, by which granzyme B, rather than caspases, cleaves GSDMC for pyroptotic induction and provides a new strategy for the therapeutic treatment of melanoma by lead compound DdBIC.
Link: https://www.nature.com/articles/s41392-025-02528-w